
Recently, the Ker Yu’s research team from the School of Pharmacy, Fudan University, published an online research paper titled “Tissue factor promotes TREX1 protein stability to evade cGAS-STING innate immune response in pancreatic ductal adenocarcinoma” on the journal Oncogene, revealing for the first time the mechanism by which TF in PDAC evades the cGAS-STING innate immune response by stabilizing TREX1 protein, and proposing the potential application of TF-inhibition therapy in the treatment of PDAC.

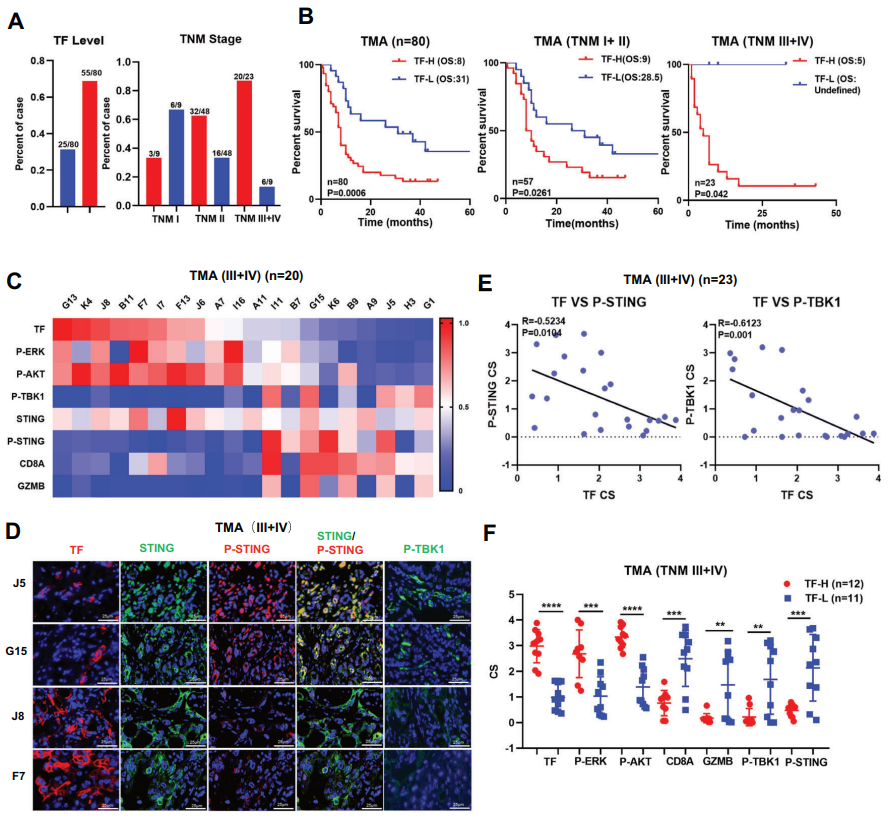
The study established the relationship between TF and cGAS-STING by analyzing the genomics and tissue microarrays of patients. In patients with TF-overexpression, the levels of STING signal activation markers P-STING/P-TBK1 were extremely low, as well as the levels of ISGs and immune effect cells were reduced. Furthermore, by constructing mouse and human pancreatic orthotopic tumor models, treatment of tumors with TF-KO/shRNA or anti-TF antibody HuSC1-39, significant anti-tumor effects were achieved and greatly promoted STING signal activation in the tumors.
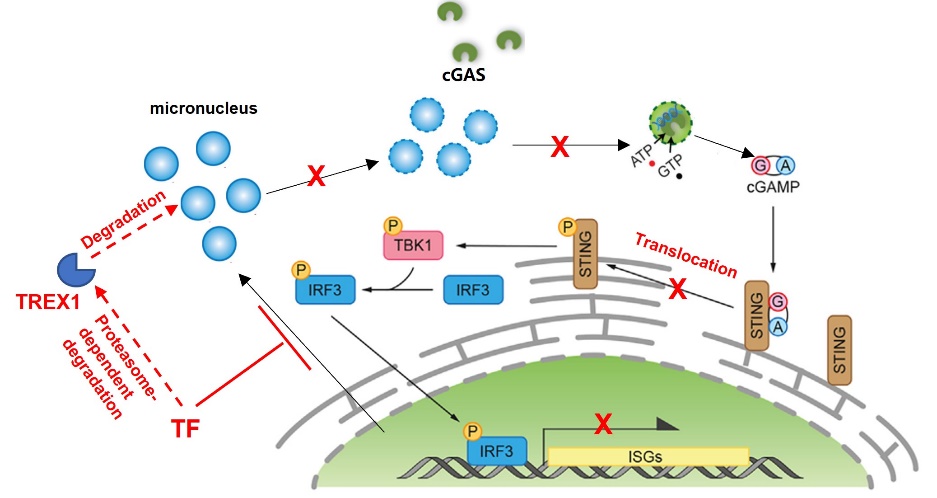
Cytosolic micronuclei accumulation is identified a classical mechanism known to trigger the cGAS-STING innate immune responses. However, poorly understood about how this regulatory mechanism is inactivation in tumor development. This study revealed that TF-overexpression can significantly inhibit the accumulation of cytosolic micronuclei and disable the cGAS-STING sensing pathway. TF-inhibition triggers cytosolic micronuclei/cGAS accumulation, initiates STING phosphorylation and STING/IRF-dependent CCL5/CXCL10 production.
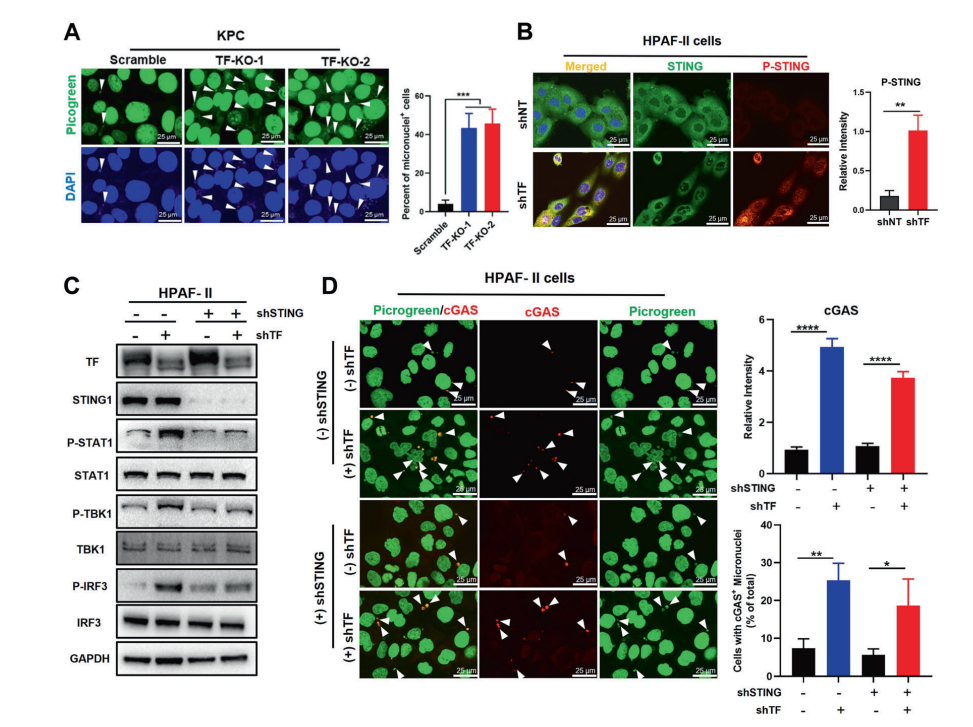
TREX1 is an exonuclease that has recently been shown to suppress innate immune responses in the tumor immune environment.This study found that TF-inhibition therapy can rapidly trigger the protein degradation of TREX1, thereby restoring micronucleus accumulation and the phosphorylation and functional activity of STING/TBK1 cascade.This study identified TF as an important immune escape regulator in PDAC. TF-inhibition therapy will provide an innovative therapy that can promote anti-tumor immune microenvironment remodeling and therapeutic effect.
Yinyin Xue, a doctoral student from the Class of 2019 at Fudan University's School of Pharmacy, is the first author of this article, and Professor Ker Yu is the corresponding author. This research has been funded by the National Natural Science Foundation of China, major projects of the National Science and Technology Support Program, and the Transformation of Scientific and Technological Achievements Fund.
Original Link: https://www.nature.com/articles/s41388-024-03248-1