
Recently, Professor Jianxin Wang's group at the School of Pharmacy, Fudan University, successfully developed an antigen self-presenting dendrosome nanovaccine. Through the synergistic co-delivery of antigens and STING agonists, this novel vaccine substantially enhances the efficacy of cancer immunotherapy. The study was published in Biomaterials entitled "Antigen Self-Presenting Dendrosomes Swallowing Nanovaccines Boost Antigens and STING Agonists Codelivery for Cancer Immunotherapy."
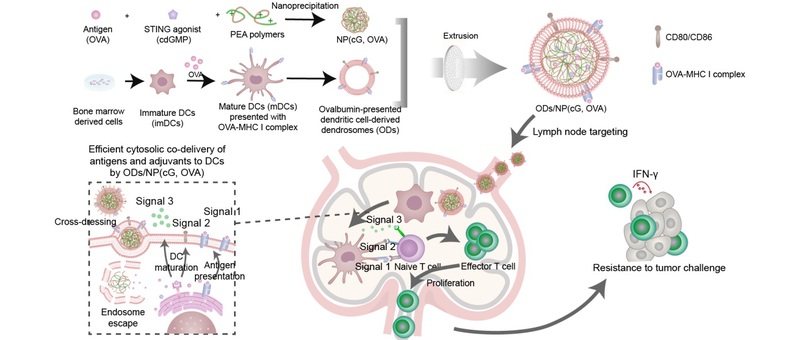
The schematic illustration of the preparation and in vivo drug delivery of dendrosomes swallowing nanovaccines ODs/NP(cG,OVA).
Cancer vaccines hold promising potential by inducing tumor-specific cytotoxic T lymphocyte (CTL) responses. Effective co-delivery of antigens and adjuvants to the cytoplasm of dendritic cells (DCs) is essential for the induction of antitumor immunity. However, tumor antigens and STING agonists, such as cyclic di-GMP (cdGMP), are prone to degradation within endosomal/lysosomal compartments, leading to suboptimal cytoplasmic delivery efficiency and inadequate CTL responses. While cationic nanocarriers can facilitate cytoplasmic delivery, their positive charge often triggers off-target effects and toxicity, negatively affecting therapeutic outcomes.
In this study, the authors designed nanoparticles based on the cationic polymer polyethylenimine (PEA), which effectively co-loaded tumor antigen peptides and the adjuvant cdGMP (NP(cG, OVA)) and encapsulated them within antigen self-presenting dendrosome vesicles (ODs). This resulted in the development of the ODs/NP(cG, OVA) nanovaccine, which enabled efficient lymph node (LN) targeting and co-delivery of antigens and adjuvants.
The results demonstrated that ODs/NP(cG, OVA) exhibited significantly reduced hepatic accumulation compared to NP(cG, OVA) and displayed enhanced targeting to LNs and DCs. ODs/NP(cG, OVA) effectively presented antigen epitopes on the ODs surface, facilitating the uptake and phagocytosis of NP(cG, OVA) by DCs. This resulted in efficient co-delivery of both antigens and adjuvants to the cytoplasm of DCs, thereby promoting antigen presentation, DC maturation, and the secretion of pro-inflammatory cytokines.
Consequently, DCs stimulated by ODs/NP(cG, OVA) effectively induced the activation, proliferation, and differentiation of antigen-specific CTLs, providing significant antitumor immunity and effectively resisting tumor invasion. This study offers an efficient vaccine strategy for cancer immunotherapy.
Postdoctoral researcher Jiaxuan Xia and doctoral student Xing Chen from the School of Pharmacy, Fudan University, contributed equally as first authors. Professor Jianxin Wang from School of Pharmacy, Fudan University and Associate Professor Junjie Pan from Huashan Hospital, Fudan University, are corresponding authors. The study was supported by grants from the National Natural Science Foundation of China (82374296, 82074277).