
Researchers Renxiao Wang and Yan Li from the Department of Medicinal Chemistry at our institute have long focused on challenging protein–protein interaction (PPI) systems as therapeutic targets. Recently, their team reported significant progress in the development of novel BAX protein agonists. Their findings were published in Journal of Medicinal Chemistry and JACS Au, both leading journals in medicinal chemistry.
BAX is a key executioner of the apoptotic pathway. Direct activation of BAX can bypass tumor resistance mechanisms associated with conventional BCL-2 inhibitors, making it a promising strategy for anticancer drug discovery. However, currently reported small-molecule BAX activators are limited in structural diversity and drug-like properties, and their activation mechanisms remain poorly understood, posing a major challenge for further development.
To identify new structural classes of BAX activators, the team conducted virtual screening against the BAX Trigger site using a library of 1.6 million compounds, successfully identifying several active hits. Through scaffold hopping and iterative structural optimization, they developed a series of compounds featuring a phenylpyrrole scaffold. Among these, compound 27c demonstrated outstanding biological performance. It binds to BAX with submicromolar affinity (IC₅₀ = 300 nM) while exhibiting weak binding to anti-apoptotic proteins such as BCL-2, BCL-XL, and MCL-1, indicating favorable selectivity.
At the cellular level, compound 27c effectively induces the conformational changes required for BAX activation, promoting its translocation from the cytoplasm to mitochondria. This leads to mitochondrial membrane depolarization, cytochrome c release, and activation of the caspase-dependent apoptotic cascade. The compound showed potent antiproliferative activity across multiple acute myeloid leukemia cell lines. Notably, its overall activity slightly surpasses that of previously reported BAX activators. In solid tumor models with high BCL-XL expression, 27c exhibited strong synergistic effects when combined with a BCL-XL inhibitor (e.g., Navitoclax), significantly enhancing cytotoxicity and supporting its potential in combination therapy.
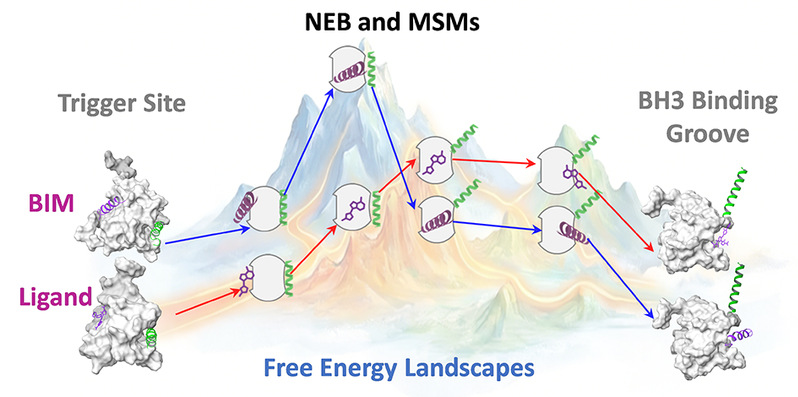
Figure 1. Structure optimization and biological characterization of phenylpyrrole-based BAX agonists.
To further elucidate the molecular mechanism of BAX activation by 27c, the team innovatively integrated the nudged elastic band (NEB) method with Markov state models (MSM), reconstructing the full activation pathway of BAX from both kinetic and thermodynamic perspectives. Comparative analysis with the natural activator peptide Bim revealed fundamental differences in their mechanisms. While both 27c and Bim bind to the Trigger site and initiate activation, their subsequent conformational pathways diverge significantly. Bim induces irreversible release of the α9 helix by overcoming a high energy barrier, thereby establishing a highly cooperative allosteric network. In contrast, although 27c rapidly triggers early conformational changes, it fails to sustain α9 helix dissociation, resulting in localized, low-cooperativity, reversible fluctuations that are insufficient to drive effective downstream oligomerization.
This mechanistic insight not only explains the activity limitations of current small-molecule BAX activators but also provides important guidance for the rational design of next-generation agonists capable of more closely mimicking natural peptide-induced activation and achieving full BAX activation.
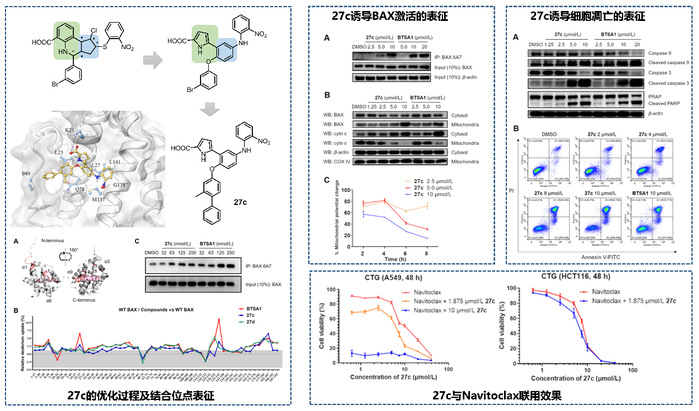
Figure 2. Comparison of the minimum free energy pathways for BAX activation induced by Bim and 27c.
These studies were jointly supervised by Professor Renxiao Wang, Associate Professor Yan Li, and Professor Lu Zhou. Doctoral student Wei Cai and master’s student Meiling Zhao contributed to the core experimental and computational work. This research was supported by the National Natural Science Foundation of China, the National Key R&D Program of the Ministry of Science and Technology, and the Science and Technology Commission of Shanghai Municipality.
Original article link:
(1)Cai W et al. Design, Synthesis, and Evaluation of Phenylpyrrole Derivatives as Small-Molecule Activators of BAX, J. Med. Chem., 2026, ASAP. https://pubs.acs.org/doi/10.1021/acs.jmedchem.5c02670
(2)Zhao, M. L. et al. Combined Nudged Elastic Band and Markov State Models Uncover Distinct BAX Activation Pathways by Bim Peptide and a Small Molecule”, J. Am. Chem. Soc. Au., 2026, 6, 1228−1237. https://pubs.acs.org/doi/10.1021/jacsau.5c01620.