
The research group led by Sun Yu at the School of Pharmaceutical Sciences, Fudan University, has recently accomplished the total syntheses of pepluanol A and epluacetal. The related findings were published online in Journal of the American Chemical Society under the title Divergent Syntheses of the Euphorbia Diterpenoids Pepluanol A and Pepluacetal.

Euphorbia diterpenoids, a class of natural products isolated from plants of the Euphorbia genus traditionally used in Chinese medicine, are prime targets in total synthesis due to their complex carbon skeletons, dense stereocenters, and diverse biological activities. In 2016, the Qiu team isolated Pepluanol A, B, and Pepluacetal from the traditional herb Euphorbia peplus, followed by the discovery of Pepluanol C and D in 2018. These compounds possess unique structures and exhibit immunosuppressive activity by targeting the Kv1.3 potassium channel, indicating potential therapeutic value for autoimmune diseases.
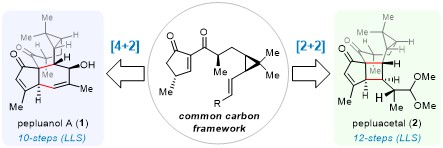
The team led by Sun Yu of our school achieved efficient syntheses of the natural products Pepluanol A and Pepluacetal by employing a divergent synthetic strategy incorporating various photochemical reactions. Specifically, the synthesis of Pepluanol A utilized a [4+2] Diels-Alder reaction as the key strategy for constructing the core skeleton, requiring only 10 steps (compared to 10-22 steps in existing routes). The synthesis of Pepluacetal employed a photoinduced [2+2] cycloaddition reaction to build the core framework, requiring just 12 steps (compared to 27 steps in existing routes). Both synthetic efforts demonstrate excellent route conciseness and precise stereochemical control.
This total synthesis research lays the foundation for subsequent biological activity studies. The project team successfully obtained a series of natural products and their analogs possessing complex structures and potential bioactivity. Through collaboration with the Yang Fan group at Renji Hospital, Shanghai Jiao Tong University, the project team confirmed that these compounds exhibit anti-pulmonary fibrosis activity. Anti-fibrotic activity tests in primary mouse lung fibroblasts showed that certain synthetic intermediates displayed potent inhibitory activity against fibronectin expression (IC50 = 3.60 μM). Notably, the natural products Pepluanol A and Pepluacetal showed weaker anti-pulmonary fibrosis activity compared to their synthetic derivatives. This work provides an important basis for the structural optimization and activity studies of this compound class, also paving the way for deeper exploration of the medicinal value inherent in these extracts from traditional Chinese medicine.
This achievement was recently published in the Journal of the American Chemical Society. Dr. Sun Yu, Principal Investigator at School of Pharmaceutical Sciences, Fudan University, is the corresponding author of the paper. Professor Yang Fan from Renji Hospital, Shanghai Jiao Tong University, is the co-corresponding author. Master's students Qiu Dongyuan and Zhao Zihan from School of Pharmaceutical Sciences, Fudan University, are the co-first authors. This work was supported by grants including the General Program of the National Natural Science Foundation of China.
Original link: https://pubs.acs.org/doi/10.1021/jacs.6c01271